Key details
Identifying new ways to detect, prevent and treat CAA
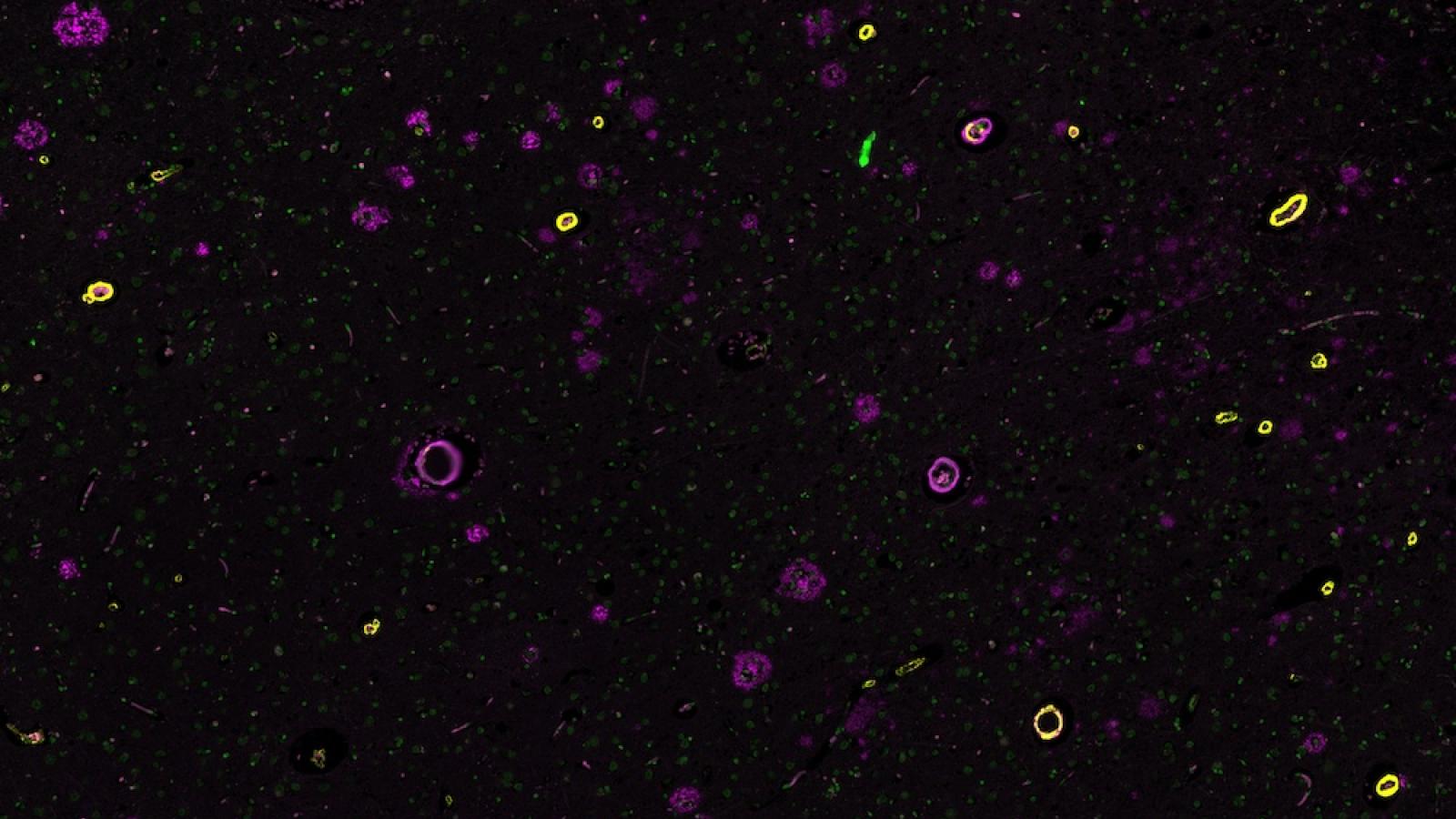
Cerebral amyloid angiopathy (CAA) is a common age-related condition, in which a sticky protein called amyloid builds up in the small blood vessels of the brain. Over time, this build-up can weaken the vessel walls and disrupt normal blood flow, increasing the risk of brain injury and dangerous bleeds. Because CAA is a major cause of disability in older adults and currently has no cure, research in this area is vital.
The van Veluw Lab aims to better understand how CAA develops and to identify new ways to detect, prevent, or treat it. The researchers combine scientific investigation with thoughtful reflection on the ethical and practical challenges of medical research. Their work also includes engaging with patients, sharing knowledge with the community, and training future scientists in a supportive, collaborative setting. By exploring new therapeutic targets and improving awareness of CAA, the team hope their findings will contribute to earlier diagnosis, safer treatments, and ultimately better outcomes for people affected by this disease.
Latest news

Prof Susanne van Veluw
Prof Susanne van Veluw is a Group Leader at the BHF-UK DRI Centre for Vascular Dementia Research. Find out more about her career and expertise on her profile page.

Research summary
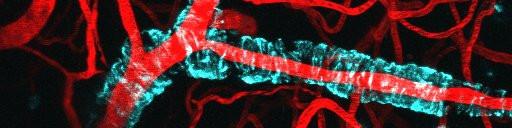
A blood vessel (in red) with amyloid-beta (in cyan) wrapped around it, in the brain of a mouse.
Mechanisms of vascular dysfunction and tissue injury in cerebral amyloid angiopathy
Cerebral Amyloid Angiopathy (CAA) is a highly prevalent cerebral small vessel disease and a major contributor to haemorrhagic stroke and vascular cognitive impairment in older adults. Despite its clinical importance, no disease-modifying therapies currently exist. Neuropathologically, CAA is defined by progressive amyloid-β (Aβ) deposition in leptomeningeal vessels, cortical arterioles, and capillaries. Advanced CAA leads to vascular dysfunction, blood–brain barrier leakage, and microvascular injury, ultimately resulting in both haemorrhagic and ischemic brain lesions. However, the molecular and cellular mechanisms linking early vascular Aβ accumulation to loss of vessel wall integrity remain poorly understood.
Recent work from the lab suggests that vascular smooth muscle cells (VSMCs) – critical regulators of arteriolar tone and low-frequency vasomotion – play a central role in early CAA pathogenesis. VSMCs are thought to contribute to perivascular Aβ clearance, and their degeneration has been associated with inflammatory changes in the perivascular space that accelerate vascular breakdown. Building on these findings, the overarching goal of this research programme is to elucidate the mechanisms that drive early vascular dysfunction in CAA, with the longer-term aim of identifying novel targets for therapeutic intervention.
To achieve this, the team combine mechanistic studies in transgenic mouse models with molecular profiling of human brain tissue. Using in vivo two-photon and wide-field optical imaging, they will examine how Aβ deposition alters VSMC structure, vasomotion dynamics, and perivascular clearance pathways. In parallel, digital spatial proteomic profiling and immunohistochemistry in post-mortem human tissue will enable them to characterise the perivascular microenvironment in vessels with and without CAA involvement. Integrating these approaches will allow the researchers to define the earliest cellular changes that precede vessel wall failure.
Ultimately, these studies aim to uncover actionable targets to enhance Aβ clearance, preserve vascular integrity, and prevent haemorrhagic and ischemic lesions in patients with CAA.
Programme Aims / Objectives
- Define early VSMC structural and functional changes induced by vascular Aβ deposition in vivo.
- Characterise perivascular clearance and vasomotion dynamics in transgenic mouse models of CAA.
- Map molecular alterations in the human perivascular environment using spatial proteomics and histopathology.
- Identify and prioritise novel therapeutic targets that could enable early intervention and prevent downstream tissue injury.
Key publications
Vacancies
Lab members
Collaborators
Lab funders
Thank you to all those who support the van Veluw Lab!