Biography
Prof David Rubinsztein is Professor of Molecular Neurogenetics and a UK DRI Group Leader at the University of Cambridge, and Deputy Director of the Cambridge Institute for Medical Research. Dr. Rubinsztein earned his MB ChB, BSc(Med)Hons, and PhD degrees from the University of Cape Town. He came to Cambridge in 1993 as a Senior Registrar in genetic pathology and was the first person to complete formal training in this field in the UK. His research is focused in the field of autophagy, particularly in the context of neurodegenerative diseases.
David's laboratory introduced and pursued the strategy of autophagy upregulation as a possible therapeutic approach for clearing toxic aggregate-prone proteins causing many various neurodegenerative diseases, and has identified drugs and novel pathways that may be exploited for this objective. He has made contributions that reveal the relevance of autophagy defects as a disease mechanism and to the basic cell biology of this important catabolic process. David was elected Fellow of the Academy of Medical Sciences (2004), EMBO member (2011), Fellow of the Royal Society (2017) and membership of Academia Europaea (2022). He was awarded the Graham Bull Prize (2007), Thudichum Medal (2017), Roger de Spoelberch prize (2017), the Goudie Medal (2020) and The Movement Disorders Research Award from American Academy of Neurology (2024).
News
Rubinsztein Lab
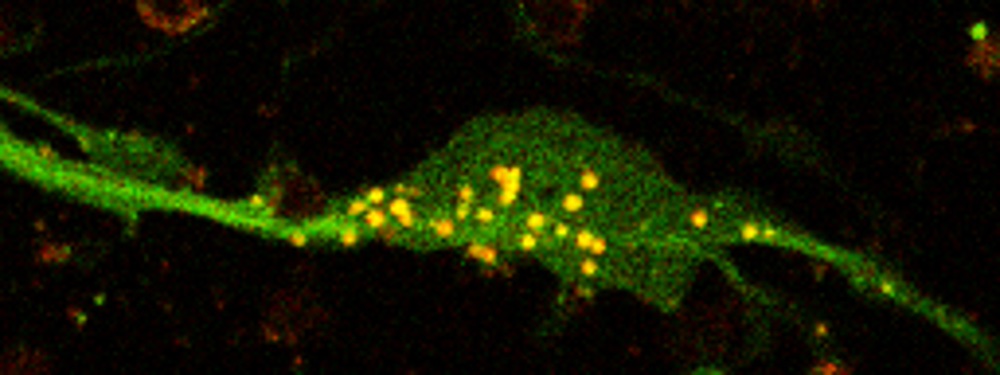
Explore the work of the Rubinsztein lab focused on deciphering the link between neurodegeneration and autophagy