Biography
Prof Bart De Strooper is a world-renowned Alzheimer's disease researcher, formerly based at the KU Leuven in Belgium, and was the founding Institute Director for the UK DRI (2017- 2023).
He was Director at the Vlaams Instituut voor Biotechnologie and led a neuroscience department of over 250 researchers. He was elected to the Academy of Medical Sciences Fellowship in 2020, and has received several awards including the Potamkin prize, the Metlife Foundation Award for Medical Research, Alois Alzheimer’s prize, the highly prestigious Brain Prize 2018 and Commander in the Order of Leopold I.
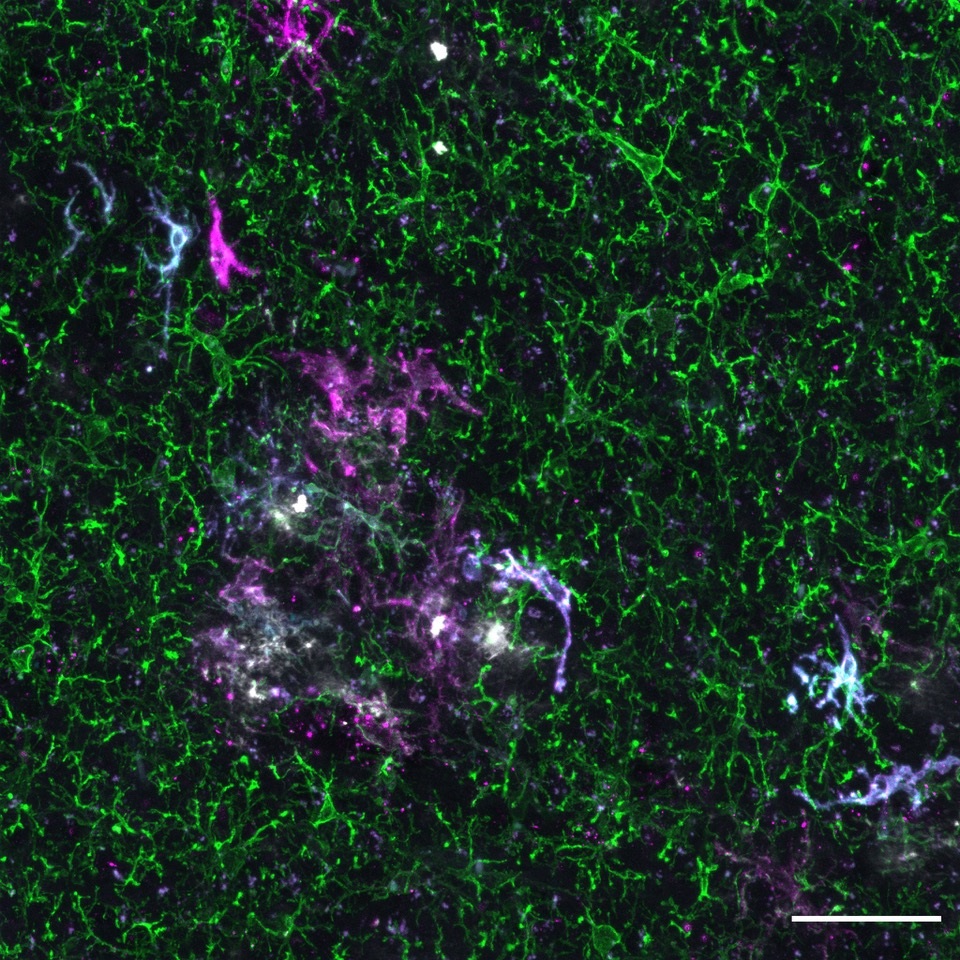
Prof De Strooper's research group at the UK DRI at UCL interrogates the fundamental mechanisms underlying Alzheimer’s and Parkinson’s disease.
De Strooper Lab
Explore the work of the De Strooper Lab Investigating the cellular reaction to amyloid beta and tau protein in Alzheimer's disease