Biography
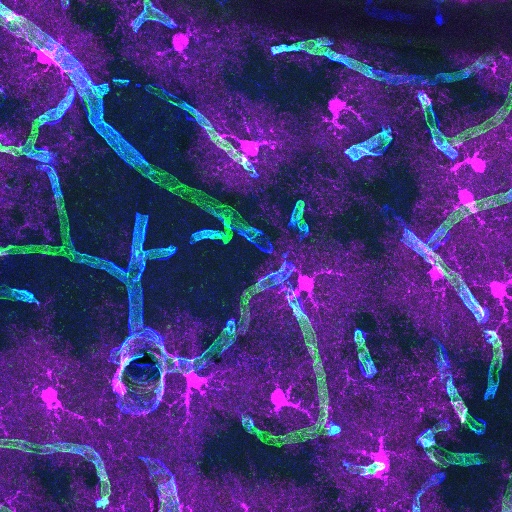
Dr Blanca Díaz Castro’s research focuses on the understanding of astrocyte-vasculature interactions and how they are important for brain function, its communication with the periphery, and the development of dementia. In particular, Dr Blanca Díaz Castro investigates the role of the astrocyte endfoot, a very specialized compartment that allows astrocytes to mediate interactions between the brain and its blood vessels, in health and diseases that lead to cognitive decline.
Dr Blanca Díaz Castro joined the UK DRI at Edinburgh in October 2019. She obtained her PhD degree at the University of Seville - Institute of Biomedicine of Seville (IBiS) and after a brief stay at Northwestern University, Chicago, she completed her postdoctoral research at University of California, Los Angeles.
Díaz Castro Lab
Explore the work of the Díaz Castro Lab, exploring the architecture, function and dysfunction of astrocyte-vasculature interactions