Biography
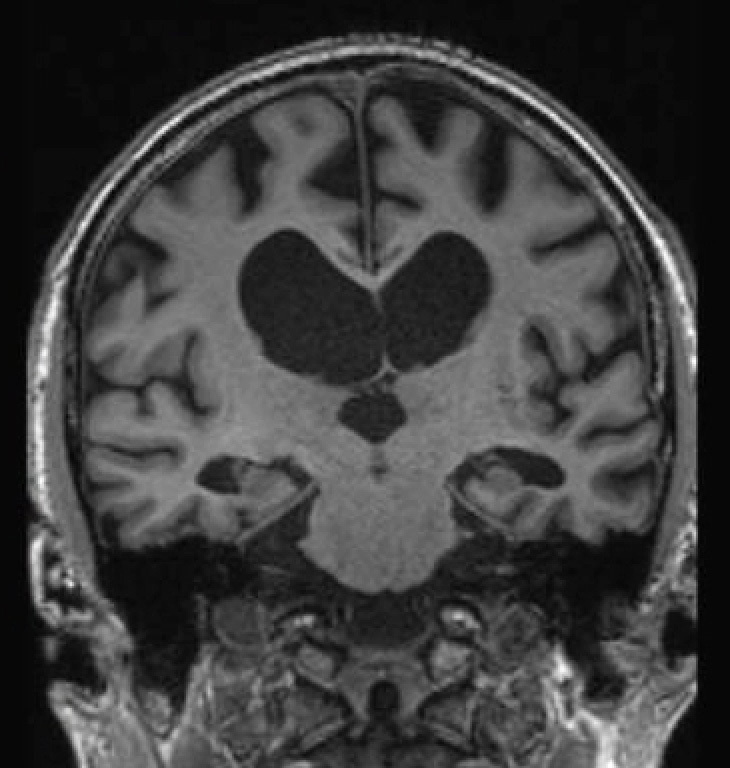
Prof Nick Fox is an expert in neuroimaging whose pioneering work in the development and application of magnetic resonance imaging (MRI) methods has had impacts on the detection, diagnosis and monitoring of progression in cognitive disorders and neurodegenerative dementias.
He is currently Professor of Neurology and Director of the Dementia Research Centre at the Institute of Neurology, University College London.
He has published over 400 papers, and is recognised as a “most highly cited” researcher by Thomson Reuters (top 1% of global citations in 2017). He has earned numerous prestigious awards, including election to the Fellowship of the Academy of Medical Sciences and NIHR Senior Investigator, and the 2017 Weston Brain Institute International Outstanding Achievement Award.
News
Key publications
Fox Lab
Explore the work of the Fox Lab, accelerating the development of effective therapies to make a difference people living with dementia