Biography
Professor Siddharthan Chandran is Director of the UK Dementia Research Institute, and an internationally leading expert in neurodegenerative diseases. Prof Chandran is a practising neurologist and scientist working at the forefront of the emerging discipline of Regenerative Neurology, renowned for his work in motor neuron disease (MND) and MS that combines laboratory and clinical research with a particular focus on human / patient stem cells for his discovery science research. Alongside his UK DRI research, Prof Chandran is Director of the Euan MacDonald Centre for Motor Neuron Disease Research and the Anne Rowling Regenerative Neurology Clinic at the University of Edinburgh. He is also a Fellow of the Royal Society of Edinburgh and the Academy of Medical Sciences.
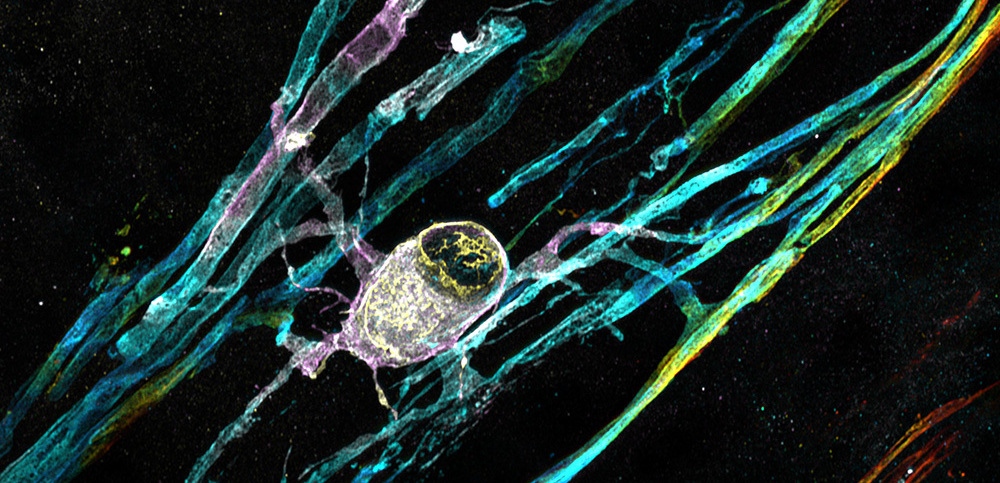
Chandran Lab
Explore the work of the Chandran Lab, Dissecting a genetic cause of ALS and FTD and identifying ways to help protect neurons.