Biography
Dr Soyon Hong received her PhD in Neuroscience in 2012 from Harvard University and completed her post-doctoral fellowship at Boston Children’s Hospital and Harvard Medical School in 2018. While in this latter role, she conducted a study that was among the first to propose microglia as critical players in synaptic pathology in disease. She now brings these expertise and knowledge to establish a new UK DRI lab group at UCL.
Statement on teaching and mentorship
"I have mentored and taught many students and technicians through lectures and laboratory settings (26 mentees in laboratory settings to date). I highly value the opportunities to mentor. It is one aspect of my career that I find truly rewarding. I believe that having a positive, stimulating environment where trainees are valued is key to having a productive and creative lab. Trainees who join my laboratory will be trained in cellular and molecular biology including neuroscience and neuroimmunology. Moreover, they will be mentored to be independent critical thinkers, a core of my teaching philosophy."
Hong Lab
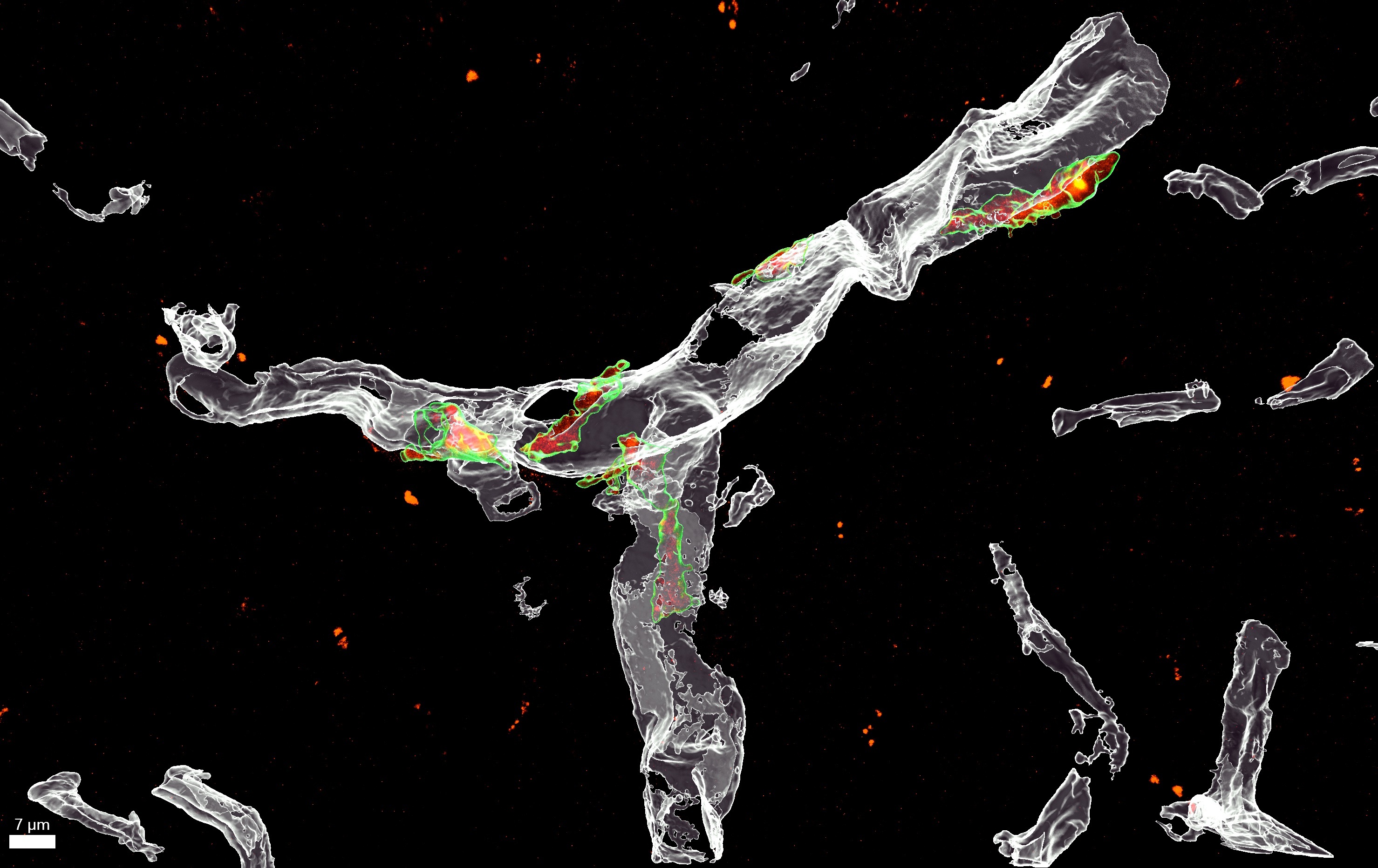
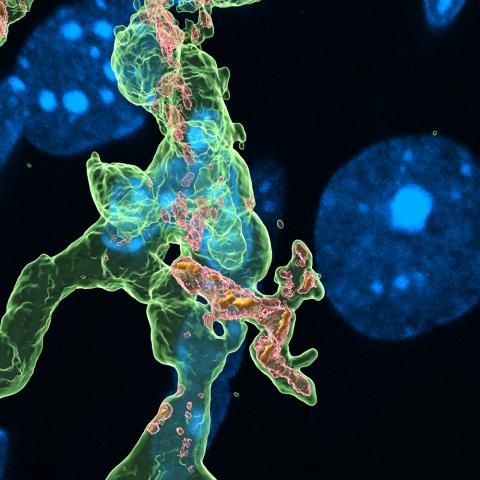
Explore the work of the Hong Lab, working to understand immune mechanisms of neural circuitry and function