Year by year, our knowledge of the fundamental biology behind dementia is improving and with it, progress made on promising new therapeutics. What has become most evident is the need to target the earliest stages of diseases like Alzheimer’s, to maximise impact and save as much of the brain as possible. A growing area of interest is the specialised blood vessel network surrounding the brain, the blood-brain barrier (BBB), whose dysfunction and breakdown is thought to be one of the initial events in several neurodegenerative diseases.
To build a better understanding and explore opportunities for treatments in this area, Director at the UK DRI’s Centre at the University of Edinburgh, Prof Giles Hardingham is taking an integrated approach, bringing together specialist researchers and clinicians, and answering major questions at the intersection between our nervous, vascular and immune systems.
The BBB, like vasculature in the rest of the body, contains a layer of endothelial cells which line the inner tube of the vessels. However, due in part to ‘tight junctions’ between these cells, the BBB is extremely selective to maintain an optimised microenvironment needed for brain function. The specialisation continues with input from multiple other cell types: pericytes which regulate blood flow; neurons that send electrical messages across the brain; astrocytes that connect blood vessels and neurons to maintain metabolism; and microglia which are the resident brain immune cells. Together these cells serve a vital collective role in protecting the brain while helping it function correctly. As Dr Blanca Díaz-Castro, an expert on astrocyte biology, explains, there are still many challenges that remain in the field.
“There are fundamental questions that we need to answer in respect to the BBB. How is the integrity of the BBB maintained? How do BBB cells interact with each other? And what molecular pathways become dysfunctional in the earliest stages of disease? I believe that solving these will provide successful avenues for early diagnosis and treatment of dementia.”
With what we now know about the complexity underlying the diseases that cause dementia, we must start to bring together different research fields and integrate our knowledge.Prof Giles HardinghamCentre Director (UK DRI at Edinburgh)
Modelling the BBB
One of the first obstacles is the development of cellular models that accurately mimic the brain and surrounding vasculature. The lab of Prof Hardingham is pioneering innovative new platforms, bringing together the multiple cell types to study them outside of the body. By using cell types from different species in the platform, the team, aided by a computer programme, are able to use small differences in DNA sequence to easily identify and separate the effects without needing to physically sort the cells.
“What we’ve discovered so far is that the interactions between cells are so important to brain health. For example, microglia need both neurons and astrocytes to maintain a healthy shape and expression of genes,” explained Prof Hardingham. “This knowledge is not only helping us build accurate research models, but also in investigating disease causes. The loss of cellular communication could result in the dysregulation that we see in advanced ageing or in disorders like Alzheimer’s.”
With funding from the Dementia Discovery Fund, Prof Hardingham and colleagues have successfully constructed a cellular model that exhibits key characteristics of the BBB including transport of molecules and impermeability to drug molecules. The platform can now be exploited to solve some of these fundamental challenges involving the BBB in health and disease, which will hopefully lead to the development of new therapeutics.
A new avenue for dementia therapies
As we age or begin to develop diseases like Alzheimer’s, there’s evidence that the BBB’s selective permeability begins to deteriorate. This leads to dysfunctional exchange or ‘crosstalk’ between the brain and the rest of the body which can lead to several complications. Perhaps most damaging is the increased influx of inflammatory mediators from the blood, stoking neuroinflammation in the brain, and perhaps driving neurodegeneration.
Related to this loss of integrity in the BBB is the dysfunction of endothelial cells that usually pack close together and form the first layer of blood vessels. This could be caused by a reduction in the number of tight junctions between the cells. The phenomenon is being investigated in cerebral small vessel disease (SVD), a condition that affects blood vessels of the brain and is a major risk factor for the development of dementia. One of the researchers at the forefront of this field is clinical vascular scientist, Prof Joanna Wardlaw CBE.
Prof Wardlaw is leading a trial (LACI-2) in people who have experienced a type of stroke often caused by damaged small blood vessels in the brain, known as a lacunar stroke. Since the endothelium is known to be damaged in small vessel disease resulting in subtle increased leakiness and damage to the brain, the team are testing the effect of two existing drugs for their endothelial cell stabilisation and support properties - cilostazol and isosorbide mononitrate currently used for other circulatory diseases. They are monitoring the effects on patient clinical outcomes including cognition and on small vessel brain damage using MRI. “If the results are encouraging, then we plan a larger trial to find new effective ways to treat small vessel disease including lacunar strokes and vascular cognitive impairment and potentially prevent some cases of dementia,” said Prof Wardlaw.
Another area of interest and possible utility for in vitro models, is in the investigation of inflammatory signalling across the BBB. When we experience an infection, the significant immune response generated impacts our brain through various routes, one of which being specific transporters and channels present in endothelial cells. The result of this can be delirium, a rapid onset but usually acute set of symptoms associated with problems thinking, focusing and memory. Although these effects usually subside as the infection is resolved, there is evidence that delirium significantly increases the risk of dementia. This would fit with our growing understanding that neuroinflammation can drive neurodegenerative processes in disease. Therefore, developing treatments to reduce this signalling across the BBB may be beneficial. This area is of particular interest at this time, as we see worrying neurological effects caused by Covid-19 infection.
Better drug design and utilising transport systems will help with drug delivery across the BBB and into the brain.Dr Axel MontagneGroup Leader (UK DRI at Edinburgh)
Helping drugs into the brain
Successfully targeting a disease mechanism is a huge achievement with the next critical step being the delivery of drugs to the right sites and cells. This is by no means a straightforward task because, as with bacteria and viruses, the BBB is very effective at preventing the entry of drug molecules developed to treat brain disorders. Prof Hardingham explains.
“In the case of some antibody treatments for brain disorders, only 0.1-1% of the drug enters the brain and where it needs to be,” explains Prof Hardingham. “That may be enough for a clinical effect in some cases but moving more drug across the BBB will allow us to lower doses, avoiding potential side effects. There are multiple approaches currently being tested but no major breakthroughs or universal platforms as of yet. This is a critical obstacle we must overcome in neurodegenerative disease therapy.”
The brain delivery methods largely fall into two categories: invasive and non-invasive. Some invasive technologies have been around for a while, such as direct injection into the cerebrospinal fluid or brain, but these are not appropriate for rollout to the wider population. Greater hope lies in the many innovative non-invasive approaches being trialled including the use of focused ultrasound to oscillate bubbles and physically open the BBB, nanoparticle systems which wrap the drug in a package capable of crossing the barrier and developments which exploit endogenous cellular transport systems such as a receptor for insulin. Dr Axel Montagne, a specialist in pericyte biology, comments on one of the more controversial topics surrounding the study of the BBB and drug delivery.
“You may assume that if the BBB had lost its integrity or become broken, it would be easier for drugs to enter the brain - some researchers are even trying to exploit this further with techniques such as ultrasound. However, I am cautious about this approach and long-term consequences of opening the BBB. I believe the key to improving diseases such as Alzheimer’s, is to repair the BBB, so it can clear the brain of protein aggregates through functional waste pathways. Better drug design and utilising transport systems will help with drug delivery across the BBB and into the brain.”
Although a number of these delivery methods look promising, they all have strengths and weaknesses, and gaining greater knowledge through these new models, will help establish successful technologies that make their way to the clinic.

The centre gather for their annual symposium at the Royal Society in 2019
On his ambitions for the UK DRI at Edinburgh Centre, which now has 12 Group Leaders, Prof Hardingham explains why the approach and strategy is laying the foundations for future treatments.
“With what we now know about the complexity underlying the diseases that cause dementia, we must start to bring together different research fields and integrate our knowledge. That’s what I’m hoping to achieve with the UK DRI Edinburgh Centre, recruiting brilliant discovery researchers and harnessing the very best clinical expertise and experience.
The foundation of success in therapeutics is good understanding of the fundamental biology and mechanisms. Not only will this help us in delivering drugs into the brain, through the development of better models for instance, but I’m also a strong believer that the dysfunction at the BBB is a key early step in multiple brain disorders and provides a promising avenue for treatments.”
Article published: 28 September 2021
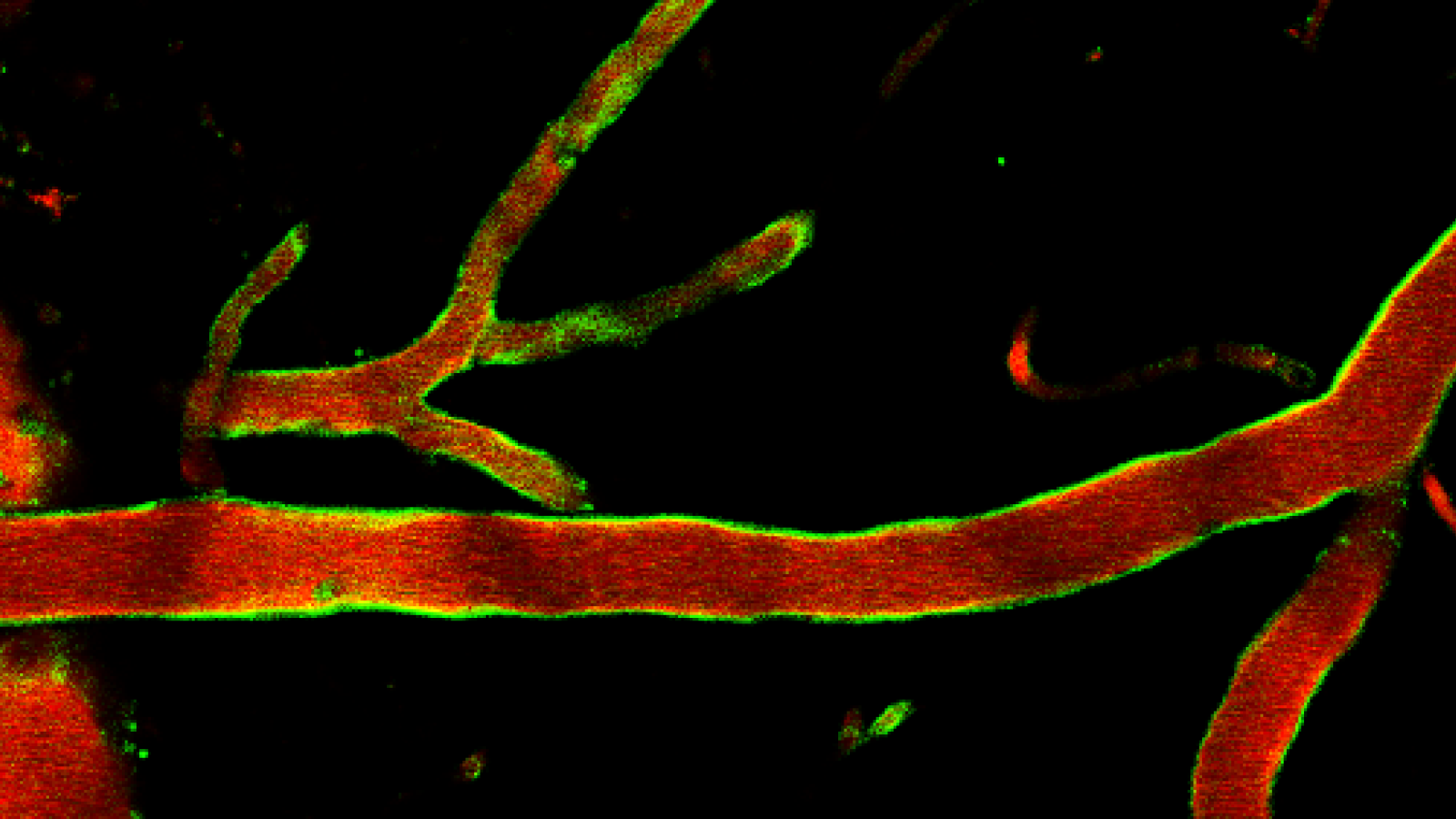
Banner image: Brain blood vessels of a mouse under a 2-photon microscope (Red = vessel marker, Green = inflamed vessels) courtesy of Dr Axel Montagne
Mixed species image: A mixed species co-culture from the lab of Prof Giles Hardingham, with mouse neurones (blue – beta-tubulin), human astrocytes (green – GFAP) and rat microglia (red – Iba1) courtesy of Dr Paul Baxter