Biography
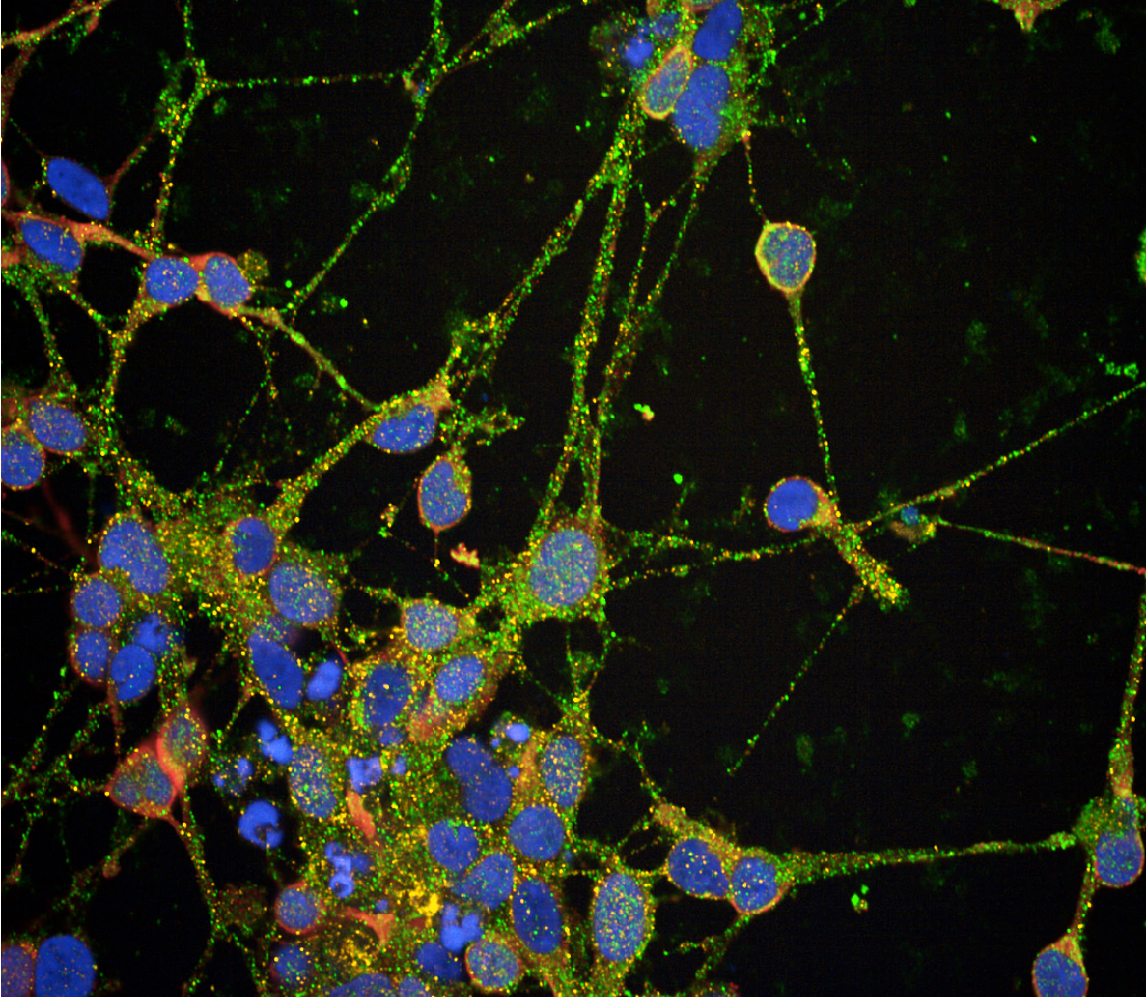
Dr Connor-Robson received her BSc in Biomedical Sciences from Cardiff University before completing her PhD on the role of the synuclein family in health and disease. In 2014, Dr Connor-Robson joined the Parkinson’s Disease Centre at the University of Oxford as a Career Development Fellow. During this time, she worked on understanding the earliest cellular pathogenic events to occur in Parkinson’s using both rodent models and iPSC-derived dopaminergic neurons. Her work highlighted the extensive roles of LRRK2 mutations in the endocytic and autophagic pathways as well as examining the role of GBA in Parkinson’s.
In 2021 Dr Connor-Robson was awarded an ARUK Research Fellowship and joined the UKDRI as an Emerging Leader and in 2024 received an ARUK Senior Research Fellowship. The work of her group now focuses on understanding the role of the endocytic pathway and polygenic risk in the development of sporadic Alzheimer’s disease using novel iPSC based models.
News
Key publications
Connor-Robson Lab
Explore the work of the Connor-Robson lab, focused on deciphering the genetics of late-onset Alzheimer's and the role of the endocytic pathway.