A team of researchers led by Dr Marc-David Ruepp, Group Leader from UK DRI at King’s College London have uncovered a new role for RNA in the development of Amyotrophic Lateral Sclerosis (ALS). Their discovery provides the molecular basis for a disease cascade that is initiated by the ALS-linked protein FUS and could serve as a platform from which to generate new therapeutics for those living with the condition.
Amyotrophic Lateral Sclerosis (ALS), or Motor Neurone Disease (MND), is a fatal condition that causes progressive weakness of the muscles due to the degeneration of motor neurones in the brain and spinal cord. The average age at onset is 60 years and death usually occurs three years after the first symptoms appear, due to respiratory failure. Although there are some treatments available that can slow the progression by a few months, those affected are in desperate need of more effective therapeutics that improve symptoms and extend life expectancy.
In 5% of ALS cases, the root cause is genetic, and scientists have identified that mutations in the FUS gene can cause an early-onset form of the disease. The FUS protein plays a crucial role in the normal functioning of our cells, binding RNA molecules to fine-tune the process that manufactures proteins from our DNA. In individuals who have mutations in the FUS gene, proteins are seen to build up in the wrong location, proving toxic to cells such as the motor neurones. However, the specific details of this process have remained elusive and until fully understood, are a barrier to the development of treatments.
Dr Marc-David Ruepp and his team, in collaboration with Prof Frederic Allain from the ETH Zürich, set about investigating the detailed biological steps behind this process in the hope that opportunities to therapeutically intervene and stop the disease may become apparent. The findings are published today (11 Dec) in the journal Nature Communications.
Using cutting-edge methods, they looked for differences in how normal and mutant FUS bind to RNA molecules in the cell and discovered an abnormal interaction with a key RNA that causes the latter’s mis-localisation and build up. These findings provide the first mechanistic and atomic resolution insights into how a toxic interaction of FUS with RNA could contribute to motor neurone degeneration, and offers hope for the generation of new treatments for individuals with the mutated form of the protein.
<p>of ALS cases, the root cause is genetic</p>
The discovery that mutant FUS undergoes toxic interactions with specific RNAs is exciting, especially because mutations in numerous RNA binding proteins are known to cause ALS. We hope that our insights will pave the way towards a better understanding of the disease and result in novel therapeutic approaches.Dr Marc-David RueppGroup Leader at UK DRI at King’s College London
Furthermore, a central component is the process was found to be shared with another genetic neurodegenerative disease, spinal muscular atrophy (SMA), that in some forms can prove fatal early in life. Therefore, this discovery may have a wider positive impact in the search for treatments for this disease.
On the next steps for the projects, first author, Dr Daniel Jutzi, commented:
“In this collaborative work, we established how exactly the FUS protein binds to central RNA molecules in the cell. Besides improving our understanding of the biological function of FUS, this knowledge will allow us to create synthetic RNAs to target FUS for therapeutic intervention.”
Prof Bart De Strooper, Director of the UK DRI, said:
“When the UK DRI was established, we were aiming to make discoveries like this that help us address the key gaps in our knowledge of disease mechanisms. We know that the neurodegenerative disorders behind dementia share several common characteristics and it’s very interesting to see another one highlighted here between ALS and SMA. Gaining a fuller understanding of these similarities, and differences, will be crucial in our approach to developing treatments.”
Reference
Article published: 11 December 2020
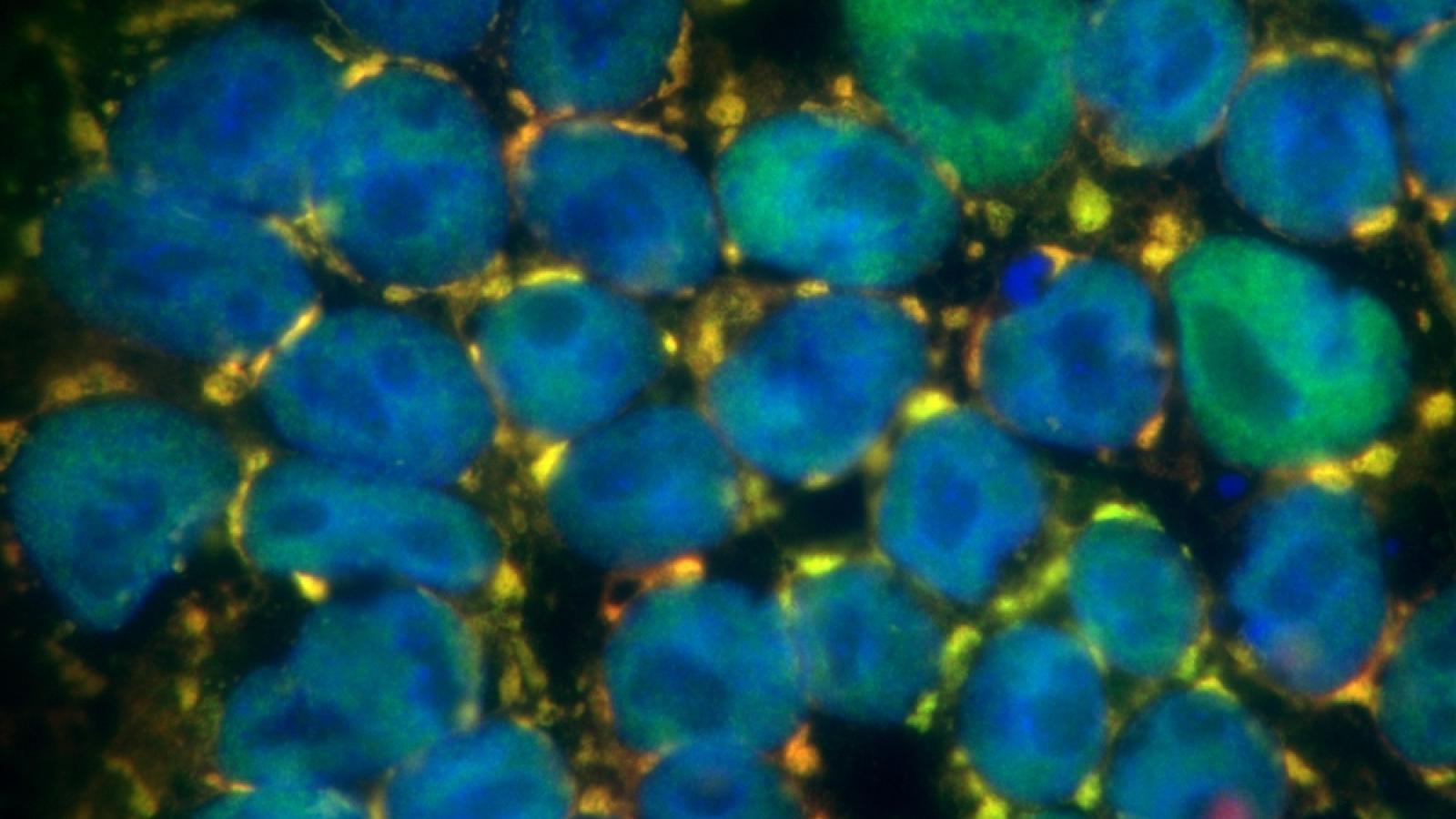
Banner image: Ruepp Lab - ALS-mutant FUS (green) forms stress granules in the cytoplasm of induced pluripotent stem cells that trap Snurportin-1 protein (red), a central component of the snRNP biogenesis machinery. Areas of yellow are due to co-localisation of both proteins. The cell nuclei are shown in blue.