Biography
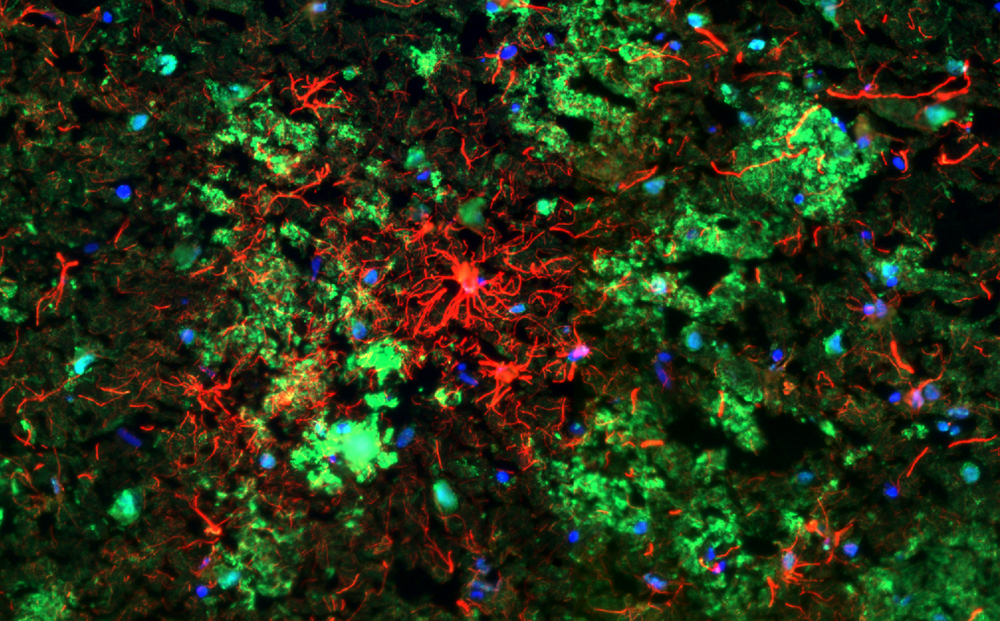
Professor B. Paul Morgan is Professor of Immunology at Cardiff University. He has worked in the complement system for over thirty years, leading a research group investigating complement biology and relevance to human disease. He has over 400 published papers and is 5th top published on 'complement system proteins' globally. Paul has made several seminal discoveries, including showing how nucleated cells recover from complement attack, the mechanism by which CD59 regulates complement pore formation and how complement triggers inflammasome activation in diverse cells. He has contributed to new understanding of MAC structure, using this knowledge to predict function. His current work focusses on roles of complement in neurodegeneration, including the development of novel anti-complement drugs for dementia (patented by Cardiff).
News
Key publications
Morgan Lab
Explore the work of the Morgan Lab, investigating exactly how key immune proteins contribute to the development of Alzheimer's disease